The Electro-Active Accommodating IOL
I just finished watching a truly mind-blowing presentation by Dr. Jay S. Pepose (Washington University School of Medicine). In it he describes an experimental intraocular lens (IOL) that changes focus from distance to near. “So what,” you say? Well, for starters, the focus of the IOL is controlled by a computer chip that is part of the IOL itself! Talk about microcomputers.
It gets even more bizarre as this tiny little computer learns an individual’s unique pupil patterns of accommodation in order to control the focus of the IOL. That’s right, in addition to focusing light (the primary role of an IOL), this little miracle of technology can detect the pupil size and learn whether the pupil is constricting from bright light or from the desire to focus at near (accommodating pupil constriction). Why is that important? Because you wouldn’t want the lens in your eye to suddenly change its focus to near every time you stepped into bright light.
This device, introduced as the Electro-Active Accommodating Intraocular Lens Implant, has no less than six different functional units – all of which must fit within the space of the eye’s natural lens. Absolutely amazing! These components include the following
- Aspheric “single piece” IOL: The “ho hum” portion of an IOL that is shared by all lenses used in modern cataract surgery. It refracts (or focuses) light onto the retina
- A thin film of liquid crystals sandwiched between a circular array of transparent electrodes within the IOL material. When a charge is placed across these crystals they form concentric rings that diffract light (similar to current multifocal IOLs). Only, because these rings are not active at all times one would expect to have fewer halos around lights with this technology.
- Two computer chips (one on either side of the optic) that control the liquid crystal display (LCD).
- Photovoltaic cells (to detect light)
- A single photocell positioned at the center of the IOL to detect light intensity
- A linear series of photocells to detect pupil diameter
- Rechargeable ceramic power cells (batteries)
- Radiofrequency Microcoils to allow for wireless inductive recharging of the
- In theory a person could recharge these IOL batteries with either a special sleep mask or pillow in which an inductive power supply was located.
Seriously, seriously cool! I half expected Dr. Pepose to end the presentation with a “and then I woke up from my dream” slide – it just seemed way too futuristic to be nearing reality. And to be fair, it will be a VERY, VERY long time before anyone in the USA ever gets to have one of these implanted within the borders of the United States. I mean, really, if the FDA can’t even approve a toric multifocal when it has been safely available everywhere else in the world for years; don’t hold your breath in anticipation of getting an Electro-Active Accommodating Intraocular Lens Implant for at least a decade.
Still, Dr. Pepose did state that the components necessary to develop this IOL are currently available. What is needed is “simply” the final engineering of the product…followed by thousands of hours and millions of dollars of wading through the tar pits of FDA bureaucracy. Oh well, it was fund to dream…
Steps in the Development of the First Electro-Active Accommodating IOL
Dr. Jay S. Pepose Presentation
I think all of us would agree that Presbyopia is the final Holy Grail for the refractive cataract surgeon and up to now we’ve had three basic approaches – pseudophakic approaches to presbyopia.
There’s multifocal IOLs but we know that they’re limited by reduce contrast sensitivity and photic phenomenon. There’s Monovision. Limited by reduced stereopsis and contrast sensitivity and Accommodating intraocular lenses, which currently are limited by insufficient and variable near vision.
So, why are we having such difficulties for the accommodating IOLs? Well, consistent performance of accommodating IOLs are influenced by intra-individual variations in capsular bag diameter, variable capsular shrinkage and fibrosis and anti-colonurgic drug affects on ciliary muscle contraction. And you see here the wide array of drugs – anti-depressants, anti-spasmodic, anti-histamines, and anti-arrythmics. It’s hard to find patients who aren’t on some or many of these medications.
So, where are we heading in the future? I think, all of us would agree if we had an intraocular lens that would consistently accommodate three diopters that would be a very popular product. Now, in 2005, a breakthrough technology occurred at the University of Arizona at their College of Optical Sciences and the development of the switchable electro-optic diffractive lens with very high efficiency for ophthalmic applications. And we’ve already seen this realized in a variety of products; the first being electro-active bi-focal (now by Pixel Optics). We’re going to see other applications in terms of slit lamps and camera technologies and now the electro-active intraocular lens, which is being developed by Elenza.
The secret sauce of this lens, so to speak, is the electrical control of the refractive index of a Nematic or Cholesteric liquid crystal which is sandwiched between a circular array of transparent electrodes and basically by changing the orientation of the liquid crystal display this changes the refractive index, as you see here, changing the focal point.
So, with an Electro-Active accommodating IOL we would want it to have:
- Fast response, as seen here in the right- Low-voltage
- High Transmission
- High Diffractive Efficiency, and we would want to have a
- Power failure-distance failsafe configuration.
Meaning, if the power were out, we’d want it to become a monofocal lens distance. So, the strategy here is to develop a Switchable Diffracted Electro-active module encapsulated within a high quality Aspheric Monofocal IOL.
- It would have Photovoltaic cells that would monitor pupil dynamics associated with accommodation.
- It would have On-board dual processors that would enable patient-specific programming because this is the accommodative effect, in terms of miosis, is very individualized.
- It would have rechargeable power cells that would last at least fifty years, switching on and off the Liquid Crystal Optics.
- It would have radiofrequency control, microcoils that would enable two-way communication and recharging externally, inductively. And have
- State of the art micro-electronics.
So, the strategy here is to use a physiological trigger taking advantage of the triad that occurs during near vision. The triad being convergence, miosis and accommodation. So, the dioptic change in the lens will be driven by pupil constriction in a 50cm vergence this would trigger a 2 diaptor impulse we would see some intermediate vision provided by miosis and the lens basically liquid crystal display would allow a three diaptor change.
Now, people dynamics were studied – 350 patients were studied, 100 were cataractous patients 100 pseudophakic patients, 50 pre and post-op and the purpose was to see, “Could an algorithm be developed that will be specific or accommodation rather than pupillary responses within regard to light itself or other stimuli?” And the patients were studied at various light levels, various distances, both Sustained and Transient Viewing and an algorithm was then developed on the initial patients and then validated on another dataset.
The conclusions of the study were as follows (four major conclusions). First the pupil response to accommodation is Nearly Universal – 99% of patient’s (in 99% of patients was demonstrated). However, there’s a tremendous amount of noise and the algorithm parameters that would differentiate the pupillary response to accommodations needed to be customized to each patient and threshold levels had to be determined. And this could now be accomplished by this application-specific integrated circuits, that would basically allow, almost like a neural net, (that would allow) the circuitry to learn each patient’s individual response and also would allow the development of screening parameters that would allow us to identify those patients who did not have a consistent pupillary response to accommodation. So those patients would not be implanted with the lens. And could be determined and screened out pre-operatively.
The lens best work in both an automated and manual mode. The automated mode, of course, working by the physiological trigger miosis. Manual mode could be it could be treated with an external wand or actually even turn on and off by specific blink sequence. This is what the lens looks like it has a central liquid crystal display. It has a Photovoltaic cell, sensing ambient light conditions. Linear photovoltaic cells that senses the pupil constriction & dilation. It has two microchips and two ceramics rechargeable power cells. It has a radio frequency microcoils for communication & recharging. So it is monofocal, in the sense, when the pupil is large, the liquid crystal optic has the same refractive index – it’s match to the monofocal optic, so the patient basically sees the distance. But, with miosis constricting the photovoltaic cells, this triggers a change in the current and the liquid crystal display orientation changes allowing +3 add.
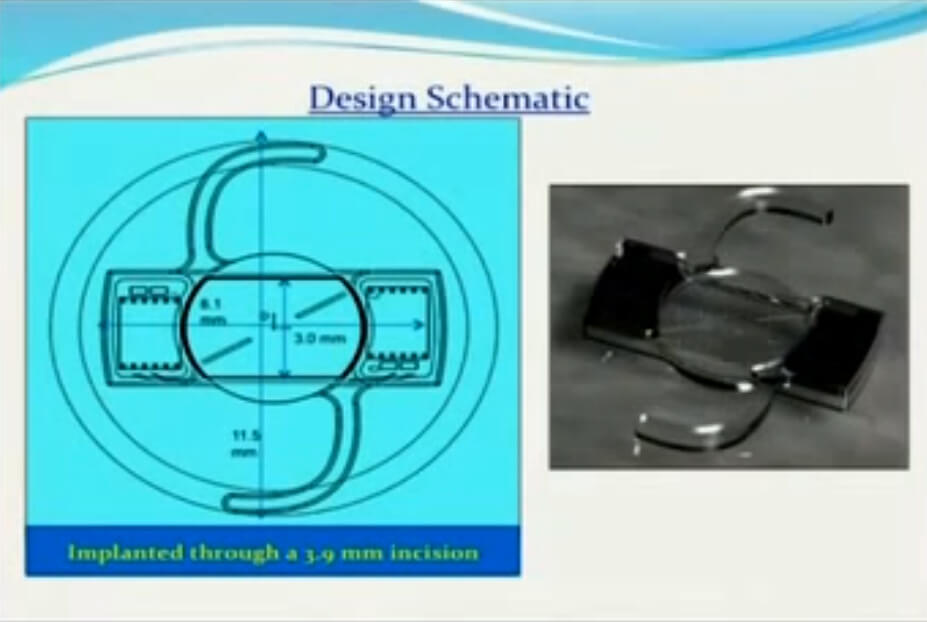
The ceramic batteries can be recharged externally by ductive recharging. You can see here it can be recharged from a sleeping eye mask, sleeping pillow or a contour neck pillow. And, currently the lenses are foldable and can be inserted through 3.9 millimeter incision. This shows the various components of the lens: the photovoltaic cells, the sensor algorithms, the microchips power cells and the microcoils.
This is the design of the lens and I would say that, it sounds like science-fiction too many but the components required to the creation of this lens are all currently available. We’re not waiting for something to be developed and while subject to many challenges, this novel IOL provides advantages, providing accommodation without movement and the technology may also have additional applications to other ophthalmic instruments and devices so the future is upon us!
Thank you for your attention.
Source: http://video.healio.com/video/





Leave a Reply